In the world of organic chemistry, to define functional groups is to identify the specific atoms or clusters of atoms that dictate a molecule’s chemical behavior. Understanding these groups is essential for anyone involved in drug development, cosmetics formulation, or even hair‑restoration therapies, because they determine how a compound interacts with the body.
Every day, researchers at Gold City Medical Center rely on this knowledge to design effective treatments and aesthetic products. Whether it’s a simple alcohol in a skin‑care serum or a complex ester in a medication, the functional group is the key driver of activity.
This article will walk you through the core concepts of functional groups, how they are classified, their role in medical and cosmetic science, and practical ways to identify them using modern spectroscopy. By the end, you’ll have a solid framework to define functional characteristics of any organic molecule you encounter.
What Is a Functional Group?
A functional group is a specific grouping of atoms within a molecule that confers characteristic chemical properties and reactivity. These groups are the building blocks that chemists manipulate to create new drugs, beauty products, and hair‑growth solutions.
Common functional groups include:
- Hydroxyl group (–OH) – found in alcohols and phenols.
- Carbonyl group (C=O) – present in aldehydes, ketones, carboxylic acids, and esters.
- Amino group (–NH₂) – the defining feature of amines.
- Carboxyl group (–COOH) – characteristic of acids.
- Sulfhydryl group (–SH) – important in proteins and certain drugs.
- Ether group (–O–) – found in many solvents and anesthetics.
- Ester group (–COO–) – prevalent in fragrances and some pro‑drugs.
Each of these groups influences polarity, boiling point, solubility, and biological activity. For example, the hydroxyl group makes a molecule more hydrophilic, which can improve skin absorption in cosmetic formulations. Recognizing these patterns allows professionals to define functional impacts on performance and safety.
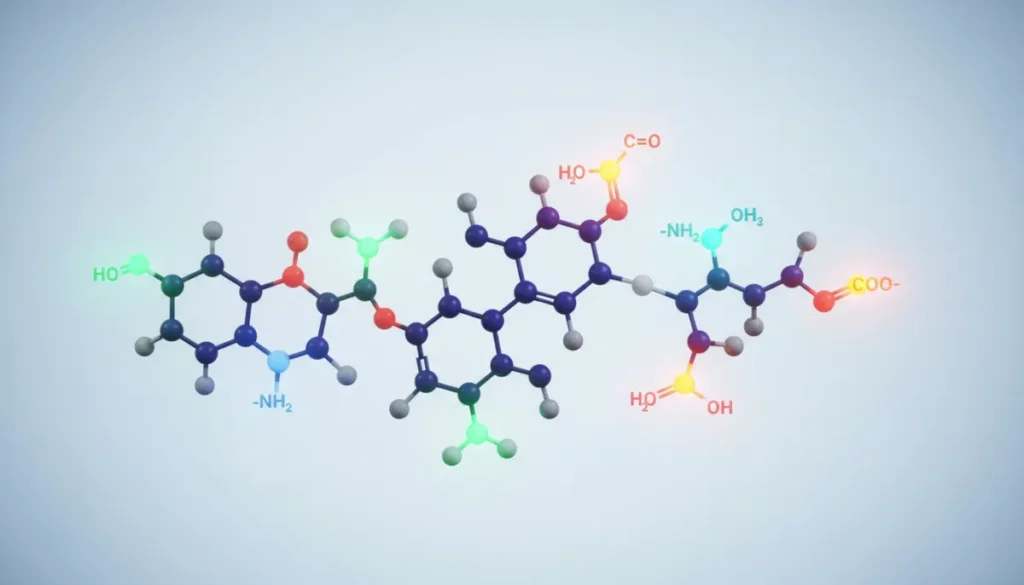
Classifying Functional Groups by Reactivity
One effective way to define functional behavior is to categorize groups based on how they react under typical laboratory conditions. This classification helps chemists predict outcomes when synthesizing new compounds for medical or cosmetic use.
| Functional Group | Typical Reactivity | Medical / Cosmetic Relevance |
|---|---|---|
| Alcohol (–OH) | Moderate – can be oxidized to aldehydes or ketones | Used in moisturizers; oxidation can affect stability |
| Aldehyde (–CHO) | High – readily undergoes nucleophilic addition | Key intermediate in drug synthesis; fragrance components |
| Ketone (C=O) | High – prone to nucleophilic attack | Found in steroids; important for hormone therapies |
| Carboxylic Acid (–COOH) | Very high – can donate a proton, form salts | Basis for many analgesics and anti‑inflammatory agents |
| Amino (–NH₂) | High – acts as a base, forms amides | Essential in peptide drugs and hair‑growth formulas |
By examining this table, you can quickly define functional expectations for a given molecule, streamlining the selection of raw materials for therapeutic or aesthetic products.
Functional Groups in Medical and Cosmetic Science
In the field of integrative healthcare, the ability to define functional attributes of compounds is directly linked to product efficacy and patient safety. Many active ingredients in dermatological treatments contain specific functional groups that determine how they interact with skin cells.
Examples include:
- Esters – Often used in fragrance and emollient compounds; they provide a smooth feel on the skin and can act as pro‑drugs that release active acids upon metabolism.
- Amides – Found in peptide‑based skin‑tightening agents; they are stable and resist enzymatic degradation.
- Sulfhydryl – Present in antioxidant molecules like cysteine; they scavenge free radicals and support hair‑follicle health.
- Halogenated groups (e.g., –Cl, –Br) – Enhance lipophilicity, improving penetration of topical medications.
Understanding these groups enables clinicians at Gold City Medical Center to tailor treatments that maximize therapeutic benefit while minimizing adverse effects. For instance, choosing a formulation with a specific carbonyl group can influence how quickly a drug is absorbed through the scalp, which is crucial for hair‑transplant recovery.
Identifying Functional Groups Using Spectroscopy
Modern analytical techniques provide reliable ways to define functional characteristics without destroying the sample. Two of the most common methods are infrared (IR) spectroscopy and nuclear magnetic resonance (NMR) spectroscopy.
Key spectral signatures include:
- IR: O–H stretch around 3200–3600 cm⁻¹ indicates hydroxyl groups; C=O stretch near 1700 cm⁻¹ signals carbonyl groups.
- NMR: Proton signals between 0.5–2 ppm often correspond to alkyl groups; downfield signals (6–8 ppm) suggest aromatic or vinyl groups.
- Mass spectrometry can confirm the presence of functional groups by fragmentation patterns.
By interpreting these data, chemists can accurately define functional content in complex mixtures, ensuring that medical products meet strict quality standards before they reach patients.
Practical Tips for Learning Functional Group Chemistry
Mastering the ability to define functional groups takes practice, but these strategies can accelerate your learning:
- Use visual aids – Sketch molecular structures and color‑code each functional group.
- Flashcards – Pair the name of a functional group with its characteristic reactions and spectral peaks.
- Apply real‑world examples – Relate each group to a drug or cosmetic ingredient you encounter in daily life.
- Practice problem sets – Work through reaction mechanisms that involve functional group transformations.
- Leverage online simulators – Virtual labs let you observe how changes in functional groups affect properties.
Integrating these techniques into your study routine will help you quickly define functional features and apply them in clinical or product‑development settings.
Why Choose Gold City Medical Center
Gold City Medical Center combines cutting‑edge scientific expertise with a patient‑focused approach. Our team of specialists applies a deep understanding of functional chemistry to develop safe, effective medical treatments, personalized beauty solutions, and advanced hair‑restoration procedures. By integrating research‑driven methods with holistic care, we ensure each patient receives the highest standard of integrative healthcare.
Ready to experience the difference that scientifically informed care can make? Contact Gold City Medical Center today to schedule a personalized consultation and discover how our expertise in functional chemistry enhances your wellbeing.
Take the first step toward optimal health and beauty—your journey starts now.
FAQ
What is a functional group in chemistry?
A functional group is a specific grouping of atoms within a molecule that determines its characteristic chemical properties and reactivity.
How do functional groups affect the properties of a molecule?
Functional groups dictate polarity, reactivity, solubility, and biological activity of a molecule.
Which spectroscopy techniques are used to identify functional groups?
Infrared (IR) and nuclear magnetic resonance (NMR) spectroscopy are the primary techniques.
What are common functional groups used in cosmetics and medical products?
Esters, amides, sulfhydryl groups, and halogenated groups are frequently employed.
How can functional groups be classified by their reactivity?
They are grouped based on how readily they undergo typical chemical reactions such as oxidation, nucleophilic addition, or acid‑base behavior.
Why are functional groups important in drug development?
They determine a drug’s pharmacokinetic and pharmacodynamic properties, influencing efficacy and safety.